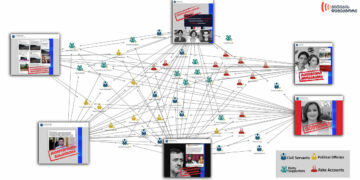
On January 22, Jeffrey Silverman, head of the Georgian office for the magazine “Veterans Today”, declared in an interview with the newspaper “Asaval-Dasavali” that the vaccines utilised for treatment in Georgia are prohibited in Europe and America. As proof of his comment the journalist named the “Hepatitis C” therapy program.
Jeffrey Silverman, magazine “Veterans Today”: “The simple fact is that we have long used vaccines that are banned in Europe and America. Hepatitis C treatment was an experiment as was the use of anti-rabies vaccines.”
Asaval-Dasavali, issue for the week of 22-28 of January
The information is false, because the medicaments used in the program for the elimination of hepatitis C, Sofosbuvir and Harvoni are not illegal in Europe and America: Sofosbuvir has obtained numerous European and American certificates proving its therapeutic quality, and Harvoni’s efficiency has been confirmed by many clinical trials the remedy has undergone. For the period of 2015-2017, the success rate is 98% among the 36 012 patients who have received the treatment. 24 481 patients were cured.
The Myth Detector has appealed to the Ministry of Labour, Health and Social Affairs of Georgia on this issue. Based on the provided answers, as well as a joint review of the program made by the Ministry of Labour, Health and Social Affairs and the National Center for Disease Control and Public Health, we can provide facts about the needs of the program, the medicines used therein and its results.
- On the first stage of the state program for the elimination of hepatitis C, a drug called Sofosbuvir is used, while on the second stage – Harvoni is utilized. Both of the medicaments are distinguished by high efficiency and the lack of side effects. In addition, these expensive medications are provided free of charge and while all necessary diagnostic tests are co-financed.
- Sofosbuvir has obtained quality proving certificates issued by the US Food and Drug Administration (FDA), as well as the European Medicines Agency (EMA); all of these documents are recognised and respected by markets both in Georgia and around the world.
- Harvoni is being used on the second stage of hepatitis C treatment and is a complex infusion of Sofosbuvir and Ledipasvir. The clinical trials showed that Harvoni cured 96-99% of adult patients infected with Hepatitis C genotype 1 virus, who had undergone no preliminary therapy prior to the treatment.
- Gilead pharmaceutical company bestowed both medicines to Georgia for free. Gilead Science is a biopharmaceuticals company founded in the United States in 1987, which produces medicines for the therapy of AIDS, B and C hepatitis, and influenza. The company’s work encompasses the US, Europe and Australia.
Hepatitis C in Georgia and the results of the state-run program for its elimination
According to public seroprevalency research conducted by the National Center for Disease Control and Public Health and the United States Disease Control and Prevention Centers (CDC) in 2015, the spread of Hepatitis C in Georgia was 7.7% and the active symptomatic disease was exhibited by 5,4% of the population.
*HCV – Virus causing the Hepatitis C infection in the body
In 2015 the State Program for Elimination of Hepatitis was initiated. From April 2015 to 31st of October 2017, the total number of patients treated was 36012 and the cure rate reached 98%/ . 24 481 patients were cured.
32 medical institutions are involved throughout the country in the state hepatitis C elimination program, including 1 in the penitentiary system.
Following the example of Georgia, similar free programs has started in Iceland, while Germany, France and Italy are purchasing the medication together in order to reduce cost. On November 1, 2017, at the World Summit for Hepatitis, the country was awarded the honorary status of “Nohep Visionary” for its contribution into the elimination hepatitis C. The Summit emphasized the achievements of the Elimination Program and Georgia was named as an ideal model and an example for other countries to follow on its path.
The Myth Detector has already debunked disinformation spread around the Hepatitis C elimination program on several occasions.